 
Two same-sign orbitals have a constructive overlap forming a molecular orbital with the bulk of the electron density located between the two nuclei. The sign of the phase itself does not have physical meaning except when mixing orbitals to form molecular orbitals. In graphical representations of orbitals, orbital phase is depicted either by a plus or minus sign (which has no relationship to electric charge) or by shading one lobe. The phase (or sign) of an orbital is a direct consequence of the wave-like properties of electrons. Two atomic orbitals can overlap in two ways depending on their phase relationship (or relative signs for real orbitals). A second condition for overlapping atomic orbitals is that they have the same symmetry. Sharing of molecular orbitals between atoms is more important when the atomic orbitals have comparable energy when the energies differ greatly the orbitals tend to be localized on one atom and the mode of bonding becomes ionic. The atomic orbital energy correlates with electronegativity as more electronegative atoms hold their electrons more tightly, lowering their energies. Because σ bonds feature greater overlap than π bonds, σ bonding and σ* antibonding orbitals feature greater energy splitting (separation) than π and π* orbitals. In MO theory molecular orbitals form by the overlap of atomic orbitals. Often even for simple molecules, AO and MO levels of inner orbitals and their electrons may be omitted from a diagram for simplicity. 
For other polyatomic molecules, an MO diagram may show one or more bonds of interest in the molecules, leaving others out for simplicity. For simple polyatomic molecules with a "central atom" such as methane ( CHĢ), a MO diagram may show one of the identical bonds to the central atom. For a diatomic molecule, an MO diagram effectively shows the energetics of the bond between the two atoms, whose AO unbonded energies are shown on the sides. The AO or MO shapes themselves are often not shown on these diagrams. Appropriate AO and MO levels are filled with electrons by the Pauli Exclusion Principle, symbolized by small vertical arrows whose directions indicate the electron spins. Degenerate energy levels are commonly shown side by side. Lines, often dashed diagonal lines, connect MO levels with their constituent AO levels. Molecular orbital diagrams are diagrams of molecular orbital (MO) energy levels, shown as short horizontal lines in the center, flanked by constituent atomic orbital (AO) energy levels for comparison, with the energy levels increasing from the bottom to the top. A mathematical description was provided by contributions from Douglas Hartree in 1928 and Vladimir Fock in 1930. Qualitative MO theory was introduced in 1928 by Robert S. 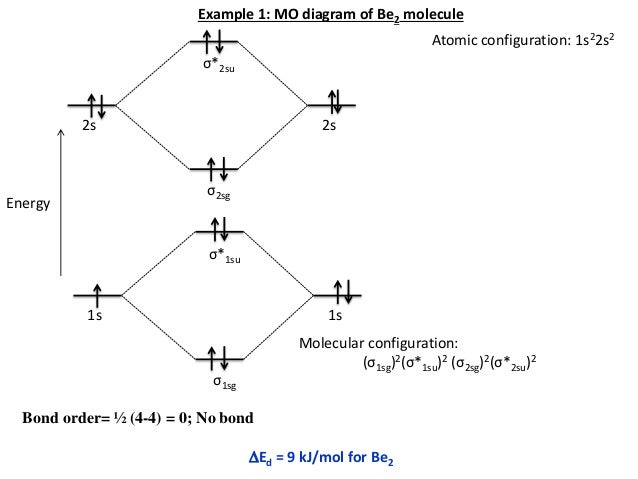
They can also predict bond strength, as well as the electronic transitions that can take place. MO diagrams can explain why some molecules exist and others do not. This tool is very well suited for simple diatomic molecules such as dihydrogen, dioxygen, and carbon monoxide but becomes more complex when discussing even comparatively simple polyatomic molecules, such as methane. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of molecular orbitals, although the electrons involved may be redistributed among the orbitals. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
